1. Follow Good Lab Practices
Cleanliness, Organization, and Accuracy are paramount when running your cannabis testing lab. We recommend that you keep your lab bench and equipment clean and organized.
-
- Always use calibrated pipettes, we recommend getting your pipettes calibrated once every 6 months (biannually)
- Use filtered tips, Filtered tips may be more expensive but it is a better option then having non-filtered tips that if contaminated, will affect future testing.
- Close pipette tip lids when not in use to reduce the risk of contamination
- Consistently wipe down lab bench, pipettes, and equipment with 10% bleach. Keeping your workspace clean and tidy will help to not only reduce contamination, but create a more efficient process for sampling.
- Use a designated area for your micro testing with designated pipettes, keeping equipment seperated is important to ensure that you are providing consistent and reproducible results.
- Transfer larger volume solutions like TSB and 70% ethanol to smaller containers to avoid contaminating full bottles. Placing your TSB or Ethanol solution into a small container ensures that if you do have contamination on your pipette, you will only contaminate that small container rather than placing a pipette into your supply container contaminating the entire supply.
- Always wear gloves and change when necessary, If you think you may have touched something that could contaminate the sample replace your gloves with a new pair. When in doubt, change your gloves.
- Stay Organized. Keep an organized lab bench with everything you need. Having everything that you need directly in front of you will ensure a efficient and timely process for testing samples. You don’t want to be running to another room to grab something you forgot.
2. Homogenize the Sample Well
Make sure your sample is well-homogenized before you weigh it out to test it for microbial contamination
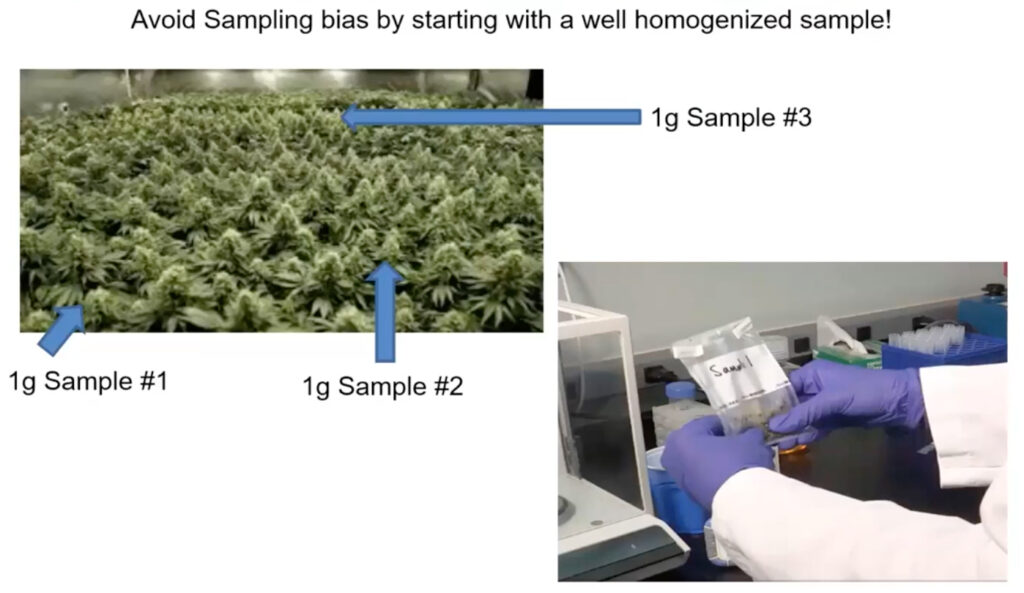
- A well homogenized sample will give a more consistent result!
- After the sample has been weighed out to test for microbial contamination in a whirl pack bag, be sure to homogenize a sample well with Tryptic Soy Broth
3. Enrich Samples for Presence Absence Tests

- No enrichment could miss the target organism when sampling with enrichment. The target organism has time to multiply, increasing likelihood it will be captured when sampling.
- Enrichment is essential when testing for pathogens that could potentially cause illness or death in immunocompromised patients.
- Enrichment greatly improves the likelihood that when you remove your 1mL for DNA purification and qPCR testing, a truly harmful pathogen will be captured. The image above visualizes the idea.
- We understand this enrichment step increases turnaround time, but it is necessary in order to properly test cannabis for pathogens and keep patients safe. Part of our mission at Medicinal Genomics is to ensure patients have access to safe, quality cannabis. It is a responsibility we take very seriously. Beware of microbial testing technologies that skip this crucial step.
4. Don’t Miss Harmful Endophytes
Endophytes are a bacterium or fungus, that lives within a plant for at least part of its life cycle without causing apparent disease. If you are surface washing you are not removing endophytes.
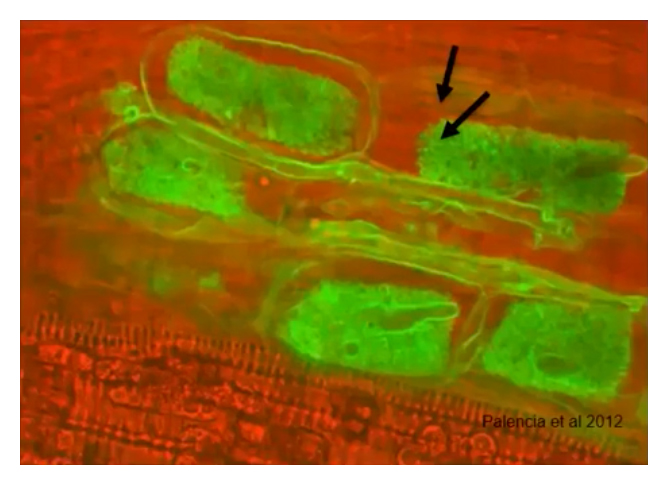
- Aspergillus sp. Can demonstrate endophytic growth.
- Complete cell lysis is the only way to get to endophyte DNA.
- Microbial pathogens can colonize many parts of the cannabis plant. Sometimes these microbes live inside the plant cells.
- Plating methods can’t detect endophytes. It is now well published that many of the harmful cannabis pathogens (Fusarium, Aspergillus etc) are endophytes. Culture based techniques need to gently soak the sample in broth so as not to kill any microbes. It is impossible to survey the endophytes inside of cannabis without lysing open cells and culture-based plating cannot detect lysed cells. As a result, culture-based platings’ achilles heal is that it cannot pick up endophytes without killing the epiphytes. qPCR can see both.
5. Use a thorough DNA Purification procedure to remove PCR inhibitors
Cannabis Flower is lipid rich, In addition, Trichomes interfere with DNA extraction techniques and Terpenes and Cannabinoids have chemical elements that interfere with reaction chemistry. Inhibitors can come into play at many parts of the polymerase chain reaction procedure.
The figure below shows the various places that inhibitors can come into play:
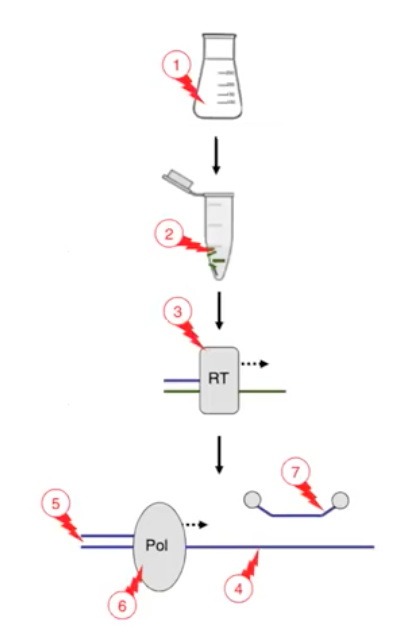
- The nucleic acids may interfere with surfaces of the vessels
- Substances may react with nucleic acids during sample processing and extraction
- Inhibitors can hamper the annealing of primers to the template
- Inhibitors can degrade, inhibit, or alter the DNA polymerase
- Substances may interfere with probe fluorophores.
- Trichomes interfere with DNA extraction techniques
- Terpenes and Cannabinoids have chemical elements that interfere with reaction chemistry
- Simple aqueous immersions DO NOT fully sample the microbial diversity of flower
- Flower is lipid-rich making it very sticky. A strong lysis buffer is needed to fully dissolve lipids, we recommend using a bead based extraction technique to yield clean DNA.
6. Automate With Liquid Handling Robotics
Automation produces More Consistent Results, Sample Tracking, LIMS Compatibility, and Saved Resources
- Great for producing reproducible results for samples (Consistency)
- Increased yield (DNA Extraction)
- More throughput and Walk-Away time for lab professionals
7. Always Confirm a Positive CQ Value
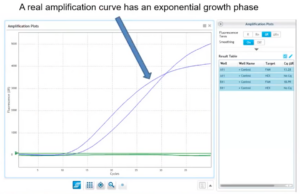
Background Trace Example
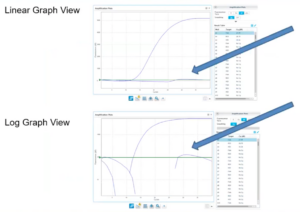
- Doesn’t have exponential growth curve
- Not a true amplification curve
8. Use Multiplexed Assays
Save time, money, and resources by testing multiple targets in a single well.
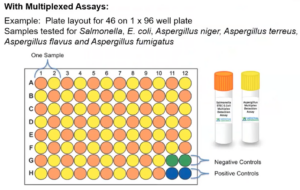
The image above shows that 46 samples can be tested for 7 pathogens on a single 96-well qPCR plate
- Previous Aspergillus multiplex assays required labs to run four species-specific assays to identify which type of Aspergillus triggered a failure. The new PathoSEEK® 5-Color Aspergillus Multiplex Assay does it all in a single qPCR reaction. This means your lab can stock fewer reagents, deliver faster results, and save money!
- Instead of testing four different species of Aspergillus individually test for all of them in on well. View Aspergillus Multiplex Assay
- Instead of testing for Salmonella and STEC E. coli individually test for both in one well. View Salmonella and STEC E. coli Multiplex Assay
- Singleplex assays require more resources and time
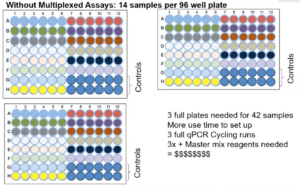
The image above shows that without multiplex assays, you can only run 14 samples per 96-well plate (42 samples total) and will need 3 qPCR plates as well as 3 qPCR runs.
9. Eliminate Dead DNA
Grim Reefer® provides a quick, accurate, and affordable solution to a problem that has haunted labs using DNA-based methods.
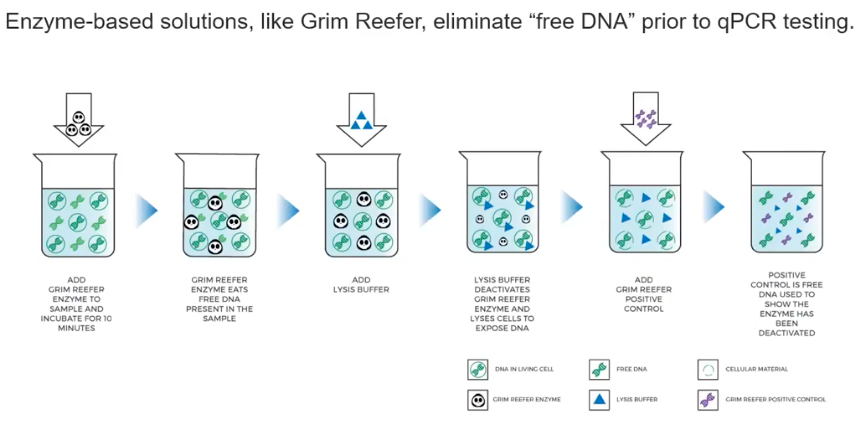
- Pro: qPCR primers and probes will amplify any DNA that matches with the target sequence
- Con: qPCR cannot distinguish between live and dead DNA
- Solution: Eliminate the contaminating dead DNA using Grim Reefer® Free DNA Removal Kit
Enzyme-based solutions, like Grim Reefer, eliminate “free DNA” prior to qPCR testing









